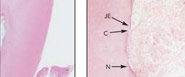
FDA clearance for True Regeneration: New PDL, New Cementum, New Bone. March 2016
Histology, LANAP, Lasers, Periodontitis
Human Clinical and Histologic Evaluation of Laser-assisted New Attachment Procedure
IJPRD Peer-Reviewed Study
Marc Nevins, DMD, MMSc
Marcelo Camelo, DDS
Peter Schupbach, PhD
Soo-Woo Kim, DMD, MS
David M. Kim, DDS, DMSc
Myron Nevins, DDS
Histology, LANAP, Lasers, Periodontitis
Histologic Evaluation of an Nd:YAG Laser Assisted New Attachment Procedure in Humans
IJPRD Peer-Reviewed Study
R.A. Yukna, R. L. Carr, G. Evans
Immediate effect of Nd:YAG laser monotherapy on subgingival periodontal pathogens: a pilot clinical study
This pilot study assessed the immediate in vivo effect of high peak pulse power neodymium-doped yttrium aluminum garnet (Nd:YAG) laser monotherapy on selected red/orange complex periodontal pathogens in deep human periodontal pockets.
LANAP, LAPIP, Lasers, Peri-Implantitis, Periodontitis
Versatile Technology Helps Provide Optimal Treatment
Inside Dentistry, November 2017, Volume 13, Number 11
LANAP, LAPIP, Lasers, Peri-Implantitis, Periodontitis
Laser Treatment for Peri-implantitis Helps Dentist Treat Patients Like Family
Sheffler uses the LANAP and LAPIP protocols for consistently excellent results
Inside Dentistry, July 2017, Volume 13, Issue 7
LANAP, Periodontitis, True Regeneration
First and Only FDA Clearance for True Regeneration
Compendium, September 2016, Volume 37, Issue 8
Just this year, the PerioLase MVP-7 became the only 510(k) Food and Drug Administration-cleared device—in dental and medical fields—that can make the claim of attachment-apparatus regeneration. – See more at: http://www.dentalaegis.com/cced/2016/09/worlds-firstand-onlyfda-clearance-for-true-regeneration#sthash.obbmAtqR.dpuf
The LANAP Protocol and Orthodontics: Offering Patients Hope Through Collaborative Treatment Plans
Todd McCracken, DDS, MALD
Today’s FDA
May/June 2014